Cisplatin induced cerebral sinus venous thrombosis in cervical
cancer patients treated with concurrent chemoradiation: a case series
Valiyaveettil Deepthi, Jilla
Swapna , Krishna Jonnalagadda Mohan , Kollu Raja, Patil Chandrasekhar
and Gupta Ranadheer (2021)
Ciplatin induced cerebral sinus venous thrombosis in cervical
cancer patients treated with concurrent chemoradiation: a case
Series ecancer 154320
Abstract
Cisplatin is a widely used chemotherapeutic agent. Concurrent
chemotherapy with cis- platin is an important component in the management of
carcinoma cervix. The common side effects of cisplatin chemotherapy include
nausea, vomiting, dyselectrolytemia, neph- rotoxicity, etc. These side effects
are anticipated and managed during chemotherapy. Thromboembolic events are rare
complications with cisplatin. We present three cases of cisplatin related
cerebral sinus venous thrombosis (CSVT). These patients were receiving
concurrent chemoradiation for carcinoma cervix. These patients presented with
neuro- logical symptoms and were evaluated and diagnosed with CSVT. They
recovered after appropriate management. Clinicians should be aware that CSVT,
though uncommon, is a life threatening
complication during cisplatin chemotherapy which should be appropri- ately
evaluated and effectively managed.
Keywords: carcinoma cervix, cisplatin, radiation, CSVT
Introduction
Cisplatin is one of the most potent alkylating antineoplastic
agents which are widely used for the treatment of several malignancies
including cervical cancer. Concur- rent chemotherapy with cisplatin during
radiation has shown better local control and overall survival compared to only
radiation in cervical cancer trials.
Cisplatin has been linked to various toxic side effects including
nausea, nephrotoxicity, cardiotoxicity, hepatotoxicity and neurotoxicity. Renal
and gastric toxicities are the most common and anticipated side effects of
cisplatin. Thromboembolic events (TEEs) is not a well-known toxicity associated
with cisplatin but has been reported in the literature. A study reported that
18.1% of cisplatin-treated cancer patients developed TEEs
We present three patients who developed cerebral sinus venous
thrombosis (CSVT) while on concurrent chemotherapy with cisplatin during the
treatment for cervical cancer.
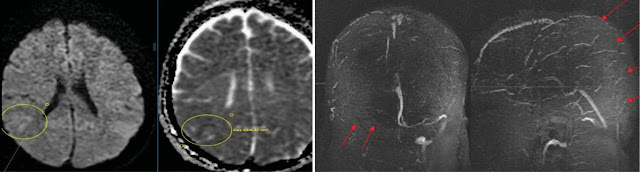
(a and b):Diffusion-weighted imaging (DWI) and Apparent Diffusion
coefficient (ADC) images showing diffusion restriction with corresponding low
ADC (marked with circle) in right parietal lobe suggesting acute infarction.
(c): Non-contrast 3D time of flight (TOF) MR venogram depicts non-
visualisation of right transverse and sigmoid sinus – Suggestive of complete
thrombosis (red arrows). (d): Non-contrast 3D TOF MR venogram depicts non-
visualisation of posterior two third superior sagittal sinus – Suggestive of
complete thrombosis (red arrows).
Case summaries
Summary 1
A 49-year-old female patient with no known co-morbidities was
diagnosed with carcinoma cervix stage IIB. She received external beam
radiotherapy to pelvis along with three cycles of concurrent chemotherapy with
cisplatin at 40 mg/m2. After 12 days of last chemo- therapy, she presented to
the emergency department with complaints of fall at home, tongue bite and loss
of consciousness. She was evaluated with Computed Tomography (CT) of the brain
which showed hyperdensity along superior sagittal and right transverse venous
sinuses suggesting the likely diagnosis of CSVT. The Magnetic Resonance Imaging
(MRI) of the brain showed thromboses in posterior half of superior sagittal
sinus, right transverse and sigmoid sinuses. She was managed with
anticoagulants, antiepileptics, analgesics and intravenous fluids. She
gradually improved and was discharged in stable condition. Post recovery she
completed her planned intra- cavitary brachytherapy. She is on regular
follow-up since 1.5 years with complete response.
Summary 2
A 37-year-old female with no known co-morbidities was diagnosed
with carcinoma cervix stage IVA. She received external beam radio- therapy to
pelvis along with four cycles of concurrent chemotherapy with cisplatin at 40
mg/m2. She developed right hemiparesis, 9 days after last cycle of cisplatin.
On evaluation, the MRI of the brain showed thrombosis in posterior part of
superior sagittal sinus, right transverse and sigmoid sinus. She was managed
with anticoagulants, antiepileptics and intravenous fluids. She improved
clinically and was discharged. Post recovery she completed the remaining course
of radiation treatment. Now she is on regular follow-up since 1.5 years with
complete response.
Summary 3
A 47-year-old female with no known co-morbidities was diagnosed
with carcinoma cervix stage IIB. She received external beam radiotherapy to
pelvis along with four cycles of concurrent chemotherapy with cisplatin at 40
mg/m2. During the course of radiation (post 7 days of fourth cycle
chemotherapy), she presented to the hospital with complaints of right upper
limb weakness. She was evaluated with the MRI of the brain which showed
anterior superior sagittal sinus and bilateral superficial cortical venous
thrombosis along with subarachnoid haemor- rhage . She was managed with
anticoagulants, antiepileptics and intravenous fluids. She recovered clinically
and completed planned radiation treatment. She is on follow-up and disease-free
since 1 year.
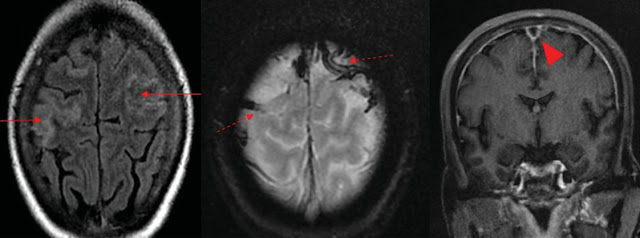
(a): Axial Fluid Attenuated Inversion Recovery (FLAIR) image
depicting mild gyral oedema in bilateral high frontoparietal lobes (red
arrows). (b): Gradient Recalled Echo (GRE) image showing blooming in cortical
veins in bilateral high frontal lobes (red dashed arrows). (c): Post contrast
T1 coronal image showing filling defect in superior sagittal sinus suggesting
thrombosis (red arrow head).
Discussion
These patients presented with neurological symptoms while on
concurrent chemoradiation for cervical cancer. They were evaluated and
diagnosed with CSVT early and were managed appropriately. The patients improved
clinically post treatment and completed the standard treatment for cervical
cancer as planned. All the three patients had no co-morbidities and no prior
history of thrombosis. Evaluation for other systemic causes was negative. The
time interval between chemotherapy and CSVT among these cancer patients
suggests a cisplatin related cause.
Cancer and cisplatin chemotherapy are well-recognised risk factors
for coagulation disorders and thrombosis. Cancer is associated with an
increased risk of venous and arterial TEEs. These events include deep venous
thrombosis, pulmonary embolism, cerebrovascular accident and unstable
angina/myocardial infarction. On an average the annual incidence rate of venous
TEE in general population is approxi- mately 117 per 100,000, whereas the
incidence in cancer patients is around one in 200 . A large cohort study
reported cancer alone increases the risk of TEE by 4.1 times and addition of
chemotherapy by 6.5 times .
CSVT during chemotherapy is rare and only few cases are reported
in literature .This condition is less common than other types of stroke. It
commonly affects large sinuses like superior sagittal sinus. Mostly no underlying
cause is identified. Around 30% of cases are attributed to inherited and
systemic inflammatory diseases. The International Study on Cerebral Venous and
Dural Sinuses Thrombosis reported 7.4% of cases of CSVT were associated with
cancer. It is more common in females with female to male ratio of 3:1.
Antineoplastic drugs like L-asparaginase and tamoxifen are established
risk factors for CSVT. Other treatment regimens with increased risk of CSVT are
(i) Folinic acid, 5fluorouracil and Irinotecan (FOLFIRI) regimen/bevacizumab
regimen in colon cancer management, (ii) concurrent chemoradiation with
temozolomide and bevacizumab in brain tumour management and (iii) cisplatin, ifosfamide, adriamycin
and vincristine regimen in Ewing sarcoma management. CSVT caused by
micrometastases from cutaneous melanoma has also been reported
The mechanism for cisplatin-induced coagulopathy is thought to be
endothelial injury activating the coagulation cascade and resulting in TEEs .
Degenerative processes of vessel walls are initiated which eventually causes
occlusive vascular disease. There is evidence regard- ing excess of myocardial
infarctions, arterial hypertension and cerebral strokes in these patients.
These complications have been reported in several malignancies but germ cell
tumours on cisplatin based regimens are at higher risk. Serum lactate
dehydrogenase levels and the body surface area are important risk predictors in
these patients . Anticoagulation, treating the underlying cause, controlling
the intracranial haemorrhage, antiseizure medication and management of focal
deficits are the main treatment strategies.
A study observed early occurrence of cardiovascular complications
secondary to cisplatin-based chemotherapy. These complications may occur during
the chemotherapy cycles or immediately after it. Karam and Koussa reported two cases who presented with cerebral
dural
sinus thrombosis while on cisplatin based chemotherapy regimen.
These patients developed neurological symptoms while on chemotherapy. MRI and
Magnetic Resonance Angiography (MRA) brain findings in these patients were
suggestive of CSVT. They were managed with anti- coagulants and supportive
care. Evaluation for other causes of CSVT was negative. They concluded that the
development of CSVT in these patients was chemotherapy related. Yamada et al reported a case of superior sagittal sinus
thrombosis in a 5-year-old girl treated with cisplatin and etoposide regimen
for a suprasellar germ-cell tumour. The patient presented with symptoms
post two cycles of chemotherapy.
A large retrospective analysis , done by Memorial Sloan-Kettering
Cancer Center confirms the incidence of TEEs in patients receiving
cisplatin-based chemotherapy. They included 932 patients with various cancers.
They included 39 (4.2%) patients who had uterine/cervical or vulvar cancers.
TEE was reported in 169 (18.1%) patients during treatment or within 4 weeks of
the last dose. They concluded that unac- ceptably high incidence of TEEs
(18.1%) is observed during the cisplatin-based chemotherapy for a variety of
cancers during the period of administration or within 4 weeks of completion of
treatment. They also suggested that TEE prophylaxis may be advisable for
patients receiv- ing cisplatin-based chemotherapy.
CSVT was reported in two patients who were on cisplatin based
chemotherapy for germ cell tumours . The authors suggested a cisplatin related
hypercoagulability as a leading risk factor in both these cases as other causes
for cancer related TEEs were ruled out. In one patient, they replaced cisplatin
with carboplatin in the third cycle of chemotherapy. The patient again
developed CSVT post carboplatin. This further confirms platin based
complication.
Conclusion
Cisplatin is a common and essential antineoplastic drug in the
management of several malignancies. CSVT as a complication of cisplatin che-
motherapy is uncommon and alarming. Clinicians should be aware of the potential
risk of development of this neurological side effect. Early diagnosis and
appropriate treatment are necessary for complete recovery.
Dr. Mohan Krishna
Neurologist
Follow my social pages:
Facebook
https://facebook.com/Drjmohankrishna/
Twitter:
https://twitter.com/drjmohankrishna/
Instagram
https://instagram.com/drjmohankrishna/
Linkedin
https://www.linkedin.com/mwlite/in/dr-j-mohan-krishna/
pinterest
https://in.pinterest.com/Drjmohankrishna/
#drmohankrishna #bestneurologist #Hyderabad #neurologist
#strokedoctor
#autoimmuneneurology #besttreatment
#NeurologyProblems
#Prevention #brainhealth
#neuroprevention
#neuroproblems #neurohealth #stroke
Follow more Visit:
https://jmkneurologist.com/


00 Reviews